 
Other columns show molecular geometries including the first column. The first column of geometries shows electron geometries. The above table shows basic geometries of molecules. Number of single bonds present = 4 – 2 = 2 Total number of electrons around N = 6 + 2 = 8 Number of electrons donated by hydrogen atoms = 2 x (H) Examples of Molecular Geometry Molecular Geometry of H 2O 
The geometry there is “bent or angular” because the lone electron pair needs more space than two bonding electron pair. For example, if a certain molecule is composed of two pairs of bonding electrons along with a lone pair, the molecular geometry is not linear. The geometries are determined based on the fact that lone electron pairs need more space than bonding electron pairs. Moreover, double bonds and triple bonds are considered as single bonds. In this case, lone electron pairs are not taken into account. Molecular geometry is the shape of a molecule predicted by considering only bond electron pairs. Therefore, the electron geometry = tetrahedral Number of lone electron pairs = 4 – 4 = 0 Total number of electrons around C = 4 + 4 = 8 Number of electrons donated by hydrogen atoms = 4 x (H) This gives the number of lone electron pairs present in the molecule. Deduct the number of single bonds present around the central atom from the steric number obtained above.This gives the number of electron groups present. Calculate the total number electrons around the central atom.Determine the number of electrons donated by other atoms.Determine the number of valence electrons in the central atom.It should be the most electronegative atom. Predict the central atom of the molecule.The following are the steps used in this determination. The electron geometry can be obtained using VSEPR theory. The electron geometry gives the spatial arrangement of all the bonds and lone pairs of a molecule. These electron pairs can be either bonding electrons or non-bonding electrons. The VSEPR theory states that electron pairs located around a certain atom repel each other. Key Terms: Electron Geometry, Lone Electron Pair, Molecular Geometry, VSEPR Theory What is Electron GeometryĮlectron geometry is the shape of a molecule predicted by considering both bond electron pairs and lone electron pairs. What is the Difference Between Electron Geometry and Molecular Geometry The main difference between electron geometry and molecular geometry is that electron geometry is found by taking both lone electron pairs and bonds in a molecule whereas molecular geometry is found using only the bonds present in the molecule. Molecular geometry can be determined by the number of bonds that a particular molecule has. Electron geometry includes the lone electron pairs present in a molecule. The VSEPR theory (Valence Shell Electron Pair Repulsion theory) can be used to determine the geometries of molecules. The geometry of a molecule can be given as either the electron geometry or the molecular geometry. The geometry of a molecule determines the reactivity, polarity and biological activity of that molecule. The F axial-I-F axial is actually 168°.Main Difference – Electron Geometry vs Molecular Geometry Due to repulsion between the axial F atoms and both the lone pair and double bond, we should expect the F-S-F bond angles to be compressed. With the more repulsive lone pair and the strongest equatorial repulsive force being between the double bond and lone pair, we should expect the F equatorial-I-O bond angle to be less than the 120° angle expected for the parent geometry ( it is actually much less, at 98°). This results in a seesaw molecular geometry. The lone pair and double bond are most repulsive, and should occupy the less crowded equatorial positions rather than the more crowded axial positions. There is one lone pair, a double bond to O, and three single bonds to F atoms around the central I atom. This molecule has five electron groups (steric number 5) with an approximately trigonal bipyramidal electronic (parent) geometry. The molecular geometry is a distorted tetrahedron. 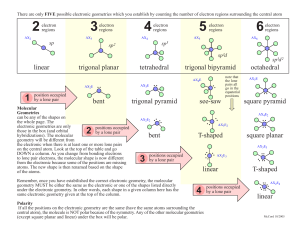
As a general rule, lone pairs are slightly more repulsive than multiple bonds, and so we might expect the O-Xe-F bond angles to be 109.5° (and the actual bond angle is 120°). VSEPR theory predicts F-Xe-F bond angles of 90°. The result is a square pyramidal molecular geometry. The double bond and lone pair will be directly opposite to each other, designated as axial positions. There is a double bond to O and a lone pair, both of which are more strongly repulsive than the single bonds to F. This molecule has six electron groups around the central Xe atom (steric number 6), and thus has an approximately octahdral electronic (parent) geometry. Use VSEPR theory to predict the geometries and draw the structures of the following. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
